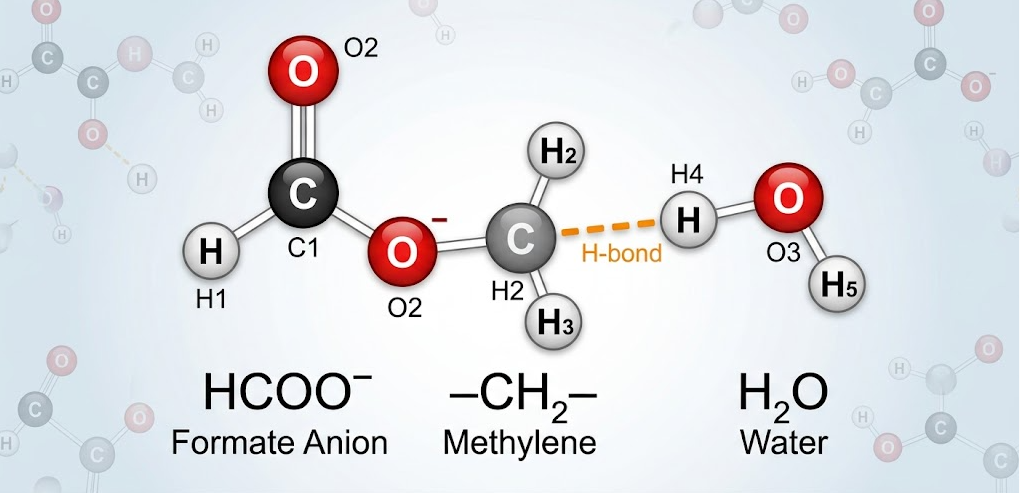
The expression hcooch ch2 h2o is not a real molecular formula. Students usually confuse it with methyl formate (HCOOCH₃) reacting with water in a hydrolysis reaction. In proper organic chemistry, methyl formate is an ester, and water breaks it down into formic acid and methanol during the reaction. This confusion happens because many notes or handwritten formulas wrongly show CH₃ as CH₂, which changes the meaning of the structure completely. CH₃ represents a methyl group, while CH₂ represents a methylene group, and both behave differently in bonding and structure. This small writing error often makes students think it is a new or unknown compound, even though it is only a common ester reaction written incorrectly. Understanding the correct structure helps students clearly see how organic molecules react and improves accuracy in solving reaction-based questions in exams and studies.
- HCOOCH₃ → Methyl formate (an ester)
- H₂O → Water (reactant in hydrolysis)
- CH₂ → Incorrect fragment in this context
The proper understanding is that CH₂ is often mistakenly written instead of CH₃. In organic chemistry, this difference is crucial because CH₃ represents a methyl group, while CH₂ represents a methylene group that behaves differently in bonding and structure.
A correct reaction representation is:
HCOOCH₃ + H₂O → HCOOH + CH₃OH
This shows the formation of formic acid and methanol, which is a classic ester hydrolysis reaction explained in many standard textbooks like Organic Chemistry by Clayden.
Why HCOOCH CH2 H2O Is Often Misinterpreted
The hcooch ch2 h2o phrase often gets misinterpreted because students read unclear handwriting, miss subscripts, and rely on simplified online notes. In many study materials, writers write the ester group CH₃ poorly, and it looks similar to CH₂, which confuses learners. Another reason for confusion comes from online explanations that remove proper chemical formatting, making real molecular structures look like unknown compounds. In reality, no separate compound called hcooch ch2 h2o exists; it is just a distorted version of an ester hydrolysis reaction. Educational chemistry highlights the importance of proper notation because even a small change can completely change the chemical identity. This is why students should always write full structural formulas instead of shorthand versions to avoid misunderstanding complex organic reactions.
Ester Hydrolysis Reaction Explained Step by Step
Ester hydrolysis is the key reaction behind hcooch ch2 h2o confusion. It is a process where an ester reacts with water to form a carboxylic acid and an alcohol.
Step-by-step breakdown:
First, the ester (such as methyl formate) is exposed to water. Under acidic or basic conditions, the ester bond begins to weaken because water acts as a nucleophile. It attacks the carbonyl carbon, leading to bond rearrangement.
Next, the molecule breaks apart, forming two products: formic acid (HCOOH) and methanol (CH₃OH). This reaction is reversible under certain conditions, meaning it can also form esters again under dehydration.
This mechanism is widely studied in organic chemistry because it demonstrates how functional groups transform through nucleophilic substitution reactions.
Role of Water (H₂O) in Organic Reactions
In reactions related to hcooch ch2 h2o, water plays a much more active role than just being a solvent. It is a key reactant in hydrolysis reactions where it helps break chemical bonds in esters. Water molecules have lone pairs of electrons that allow them to attack electrophilic centers such as carbonyl carbon atoms. This initiates the breakdown of the ester structure into simpler compounds. Without water, the hydrolysis process would not occur, which shows its essential role in organic chemistry reactions. Water is also involved in many biological and industrial processes where hydrolysis is required, making it one of the most important reactants in both natural and synthetic chemistry systems.
Common Exam Mistakes Students Make with This Formula
Students often make mistakes when interpreting hcooch ch2 h2o, mainly due to incorrect notation and lack of understanding of ester chemistry. One of the most common errors is writing CH₂ instead of CH₃, which leads to an incorrect molecular structure. Another mistake is assuming that the expression represents a completely new compound rather than a reaction process. Many students also forget to include proper hydrolysis conditions such as acid or base catalysts, which are necessary for the reaction to occur. Additionally, missing step-by-step mechanisms in answers often reduces marks in exams. These mistakes highlight the importance of understanding both correct chemical notation and reaction pathways rather than memorizing simplified formulas.
Real-World Applications of Ester Hydrolysis
Although hcooch ch2 h2o itself is not a real compound, the ester hydrolysis reaction it represents plays many practical roles in real life. One major use appears in soap production through saponification, where fats break down into glycerol and soap molecules. The food industry also uses this reaction for flavor development and for breaking down ester-based compounds. In pharmaceuticals, ester hydrolysis supports drug metabolism by helping active compounds release inside the body through breakdown reactions. It also helps recycle biodegradable plastics by breaking polymer chains into reusable components. These applications show that ester hydrolysis is not just an academic topic but an important reaction that industries and biological systems widely use.
Scientific References and Trusted Sources
The concept behind hcooch ch2 h2o appears in standard chemistry resources that focus on ester reactions and organic mechanisms. Trusted educational materials clearly explain correct molecular notation and reaction pathways to avoid confusion. Reliable chemistry references describe ester hydrolysis in detail, including reaction steps, catalysts, and product formation. Academic institutions widely use these sources to teach organic chemistry fundamentals and help students understand real molecular behavior instead of miswritten formulas. Studying verified scientific material builds a strong foundation and reduces common misconceptions in organic chemistry.
Conclusion
The expression hcooch ch2 h2o does not represent a real chemical compound but is a miswritten form of an ester hydrolysis reaction involving methyl formate and water. The correct interpretation shows that esters react with water to form carboxylic acids and alcohols through a well-known reaction mechanism. Understanding the proper molecular structure and avoiding notation errors like CH₂ instead of CH₃ is essential for accurate learning in organic chemistry. This topic highlights the importance of correct chemical writing, reaction understanding, and conceptual clarity in both academic studies and practical applications.
FAQs
1. Is HCOOCH CH2 H2O a real compound?
No, it is not a real compound. It is a miswritten representation of an ester hydrolysis reaction.
2. What is the correct formula related to this reaction?
The correct compound is methyl formate (HCOOCH₃).
3. What are the products of ester hydrolysis?
The products are formic acid (HCOOH) and methanol (CH₃OH).
4. Why is CH₂ incorrect in this formula?
CH₂ is a different chemical fragment; the correct group in this reaction is CH₃ (methyl group).
For More UpdateS Visit: Accurate Unit Convert









Leave a Reply