How to calculate formal charge is one of the most essential skills in chemistry. Whether you’re a high school student, preparing for exams, or diving deeper into molecular structures, mastering formal charge helps you predict stability, determine the best Lewis structure, and understand chemical behavior.
In this comprehensive guide, you’ll learn how to calculate formal charge from Lewis structure, explore formulas, work through real examples like carbonate ions, and understand how double bonds affect calculations. By the end, you’ll have a clear, confident grasp of the concept.
What Is Formal Charge in Chemistry?
Formal charge is a theoretical concept used in chemistry to estimate the distribution of electrons in a molecule or ion. It helps determine whether atoms in a structure are neutral, positively charged, or negatively charged.
In simple terms, formal charge answers this question:
“Does this atom have more or fewer electrons than it should?”
This concept is crucial when learning how to calculate formal charge chemistry, because it allows chemists to:
- Predict the most stable structure of a molecule
- Compare different Lewis structures (resonance forms)
- Understand reactivity and bonding
Why Learning How to Calculate Formal Charge Matters
When you draw Lewis structures, there are often multiple possible arrangements. Formal charge helps you choose the correct one.
Key reasons to learn how to calculate formal charge of an atom include:
- Identifying the most stable structure
- Minimizing charge separation
- Placing negative charges on more electronegative atoms
- Understanding resonance structures
In real-world chemistry, molecules tend to adopt structures with the lowest possible formal charges.
How to Calculate Formal Charge Formula
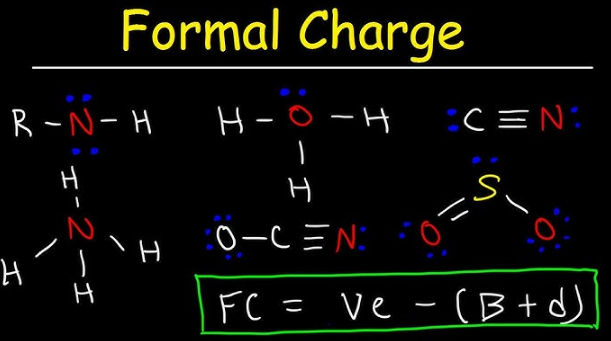
The foundation of this concept lies in the formula. Understanding how to calculate formal charge formula is the first step.
Formal Charge Equation
Formal Charge (FC) =
Valence Electrons − Nonbonding Electrons − (Bonding Electrons ÷ 2)
Explanation of Terms
- Valence electrons: Electrons in the outer shell of an atom
- Nonbonding electrons: Lone pair electrons
- Bonding electrons: Shared electrons in bonds
Simplified Version
FC = V − L − (B/2)
Where:
- V = valence electrons
- L = lone electrons
- B = bonding electrons
This is the standard how to calculate formal charge equation used in chemistry.
Also Read:- How to Cook a Ham in the Oven Perfectly Every Time
Step-by-Step: How to Calculate Formal Charge
Let’s break it down into a simple process.
Step 1: Draw the Lewis Structure
Before learning how to calculate formal charge from Lewis structure, you must correctly draw the molecule.
Step 2: Identify Valence Electrons
Check the periodic table for each atom.
Step 3: Count Lone Pair Electrons
These are nonbonding electrons.
Step 4: Count Bonding Electrons
Include all shared electrons in bonds.
Step 5: Apply the Formula
Plug values into the equation.
How to Calculate Formal Charge of an Atom (With Example)
Let’s take a simple example: water (H₂O)
- Oxygen has 6 valence electrons
- It has 4 nonbonding electrons (two lone pairs)
- It shares 4 bonding electrons
FC = 6 − 4 − (4/2)
FC = 6 − 4 − 2
FC = 0
So, oxygen has a formal charge of 0.
Hydrogen:
- Valence = 1
- Lone electrons = 0
- Bonding electrons = 2
FC = 1 − 0 − (2/2) = 0
All atoms are neutral → stable molecule.
How to Calculate Formal Charge from Lewis Structure
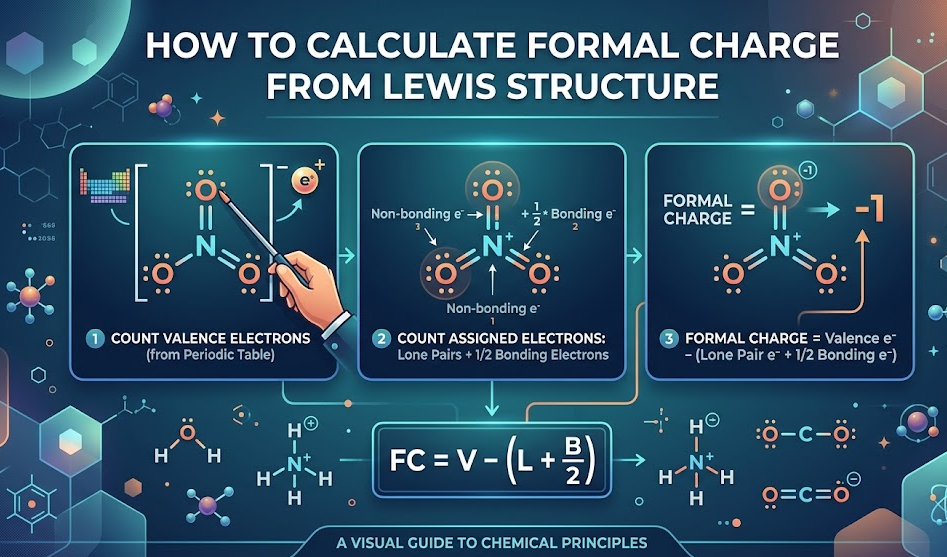
When you already have a Lewis structure, the process becomes faster.
Key Insight
You don’t need to redraw anything—just count electrons directly.
Example: Ammonia (NH₃)
- Nitrogen: 5 valence electrons
- 2 lone electrons
- 6 bonding electrons
FC = 5 − 2 − (6/2)
FC = 5 − 2 − 3
FC = 0
Hydrogen atoms = 0 formal charge
This confirms the structure is stable.
Also Read:- How to Convert Radians to Degrees (Easy Guide)
How to Calculate Formal Charge of a Molecule
When working with entire molecules, calculate the formal charge of each atom individually, then evaluate the total.
Key Rule
The sum of all formal charges must equal:
- 0 for neutral molecules
- The ion charge for ions
Example: Carbon Dioxide (CO₂)
Each oxygen and carbon atom has a formal charge of 0.
Total = 0 → Correct structure.
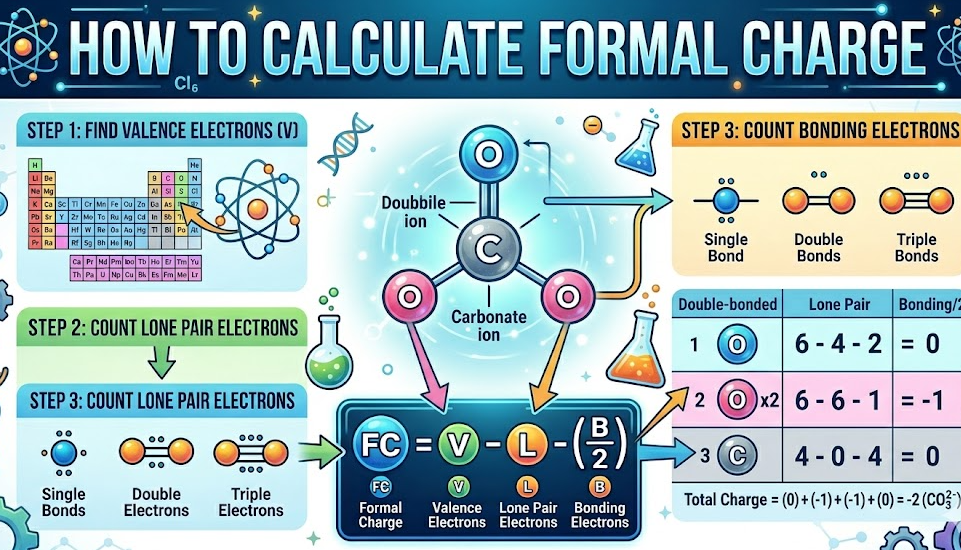
How to Calculate Formal Charge of CO₃²⁻ (Carbonate Ion)
This is a very common exam question and a perfect example of resonance.
Step 1: Draw the Structure
Carbon is central with three oxygen atoms.
Step 2: Assign Electrons
One double bond and two single bonds (in resonance forms).
Step 3: Calculate Charges
Double-bonded Oxygen
FC = 6 − 4 − (4/2) = 0
Single-bonded Oxygen
FC = 6 − 6 − (2/2) = −1
Carbon
FC = 4 − 0 − (8/2) = 0
Total Charge
−1 + −1 + 0 + 0 = −2
This confirms how to calculate formal charge of CO₃²⁻ correctly.
Also Read:- How to Repair Screen Door: Easy DIY Fix Guide
How to Calculate Formal Charge with Double Bonds
Double bonds change how bonding electrons are counted.
Important Rule
Each bond contains 2 electrons.
A double bond = 4 electrons.
Example: Oxygen in CO₂
- Valence = 6
- Lone electrons = 4
- Bonding electrons = 4
FC = 6 − 4 − (4/2) = 0
Even with double bonds, the formula remains the same—only the number of bonding electrons changes.
Common Mistakes When Calculating Formal Charge
Even experienced students make errors when learning chemistry.
Miscounting Electrons
Always double-check lone pairs and bonds.
Forgetting to Divide Bonding Electrons
This is one of the most common mistakes.
Ignoring Overall Charge
Ensure the total matches the molecule’s charge.
Choosing the Wrong Lewis Structure
Formal charge only works if the structure is drawn correctly.
Tips to Master Formal Charge Quickly
Understanding how to calculate formal charge of molecule becomes easier with practice.
Focus on Patterns
- Neutral atoms often have FC = 0
- Oxygen prefers −1 or 0
- Nitrogen prefers 0
Practice Common Molecules
- CO₂
- NH₃
- NO₃⁻
- CO₃²⁻
Use Shortcuts
You can sometimes estimate formal charge by comparing bonds vs expected valence.
Formal Charge vs Oxidation State
These two concepts are often confused.
Formal Charge
- Assumes equal sharing of electrons
- Used for Lewis structures
Oxidation State
- Assumes full transfer of electrons
- Used in redox reactions
Understanding this difference improves your grasp of equation.
Real-World Applications of Formal Charge
Formal charge isn’t just theoretical—it has practical importance.
Predicting Chemical Stability
Molecules prefer minimal charge separation.
Understanding Reaction Mechanisms
Helps identify reactive sites.
Designing Molecules
Used in pharmaceuticals and materials science.
Also Read:- How to Set Up Voicemail on Any Phone (Easy Guide)
Conclusion
Learning how to calculate formal charge is a fundamental step in mastering chemistry. By understanding the formula, applying it to Lewis structures, and practicing real examples like carbonate ions and double bonds, you can confidently determine the most stable molecular structures.
The key takeaway is simple:
Formal charge helps you verify whether a structure makes chemical sense.
With consistent practice, calculating formal charge becomes second nature—and an essential tool in your chemistry toolkit.
FAQs
What is the easiest way to calculate formal charge?
Use the formula:
Valence − Lone electrons − (Bonding electrons ÷ 2)
How do you calculate formal charge from a Lewis structure?
Count lone pairs and bonds directly from the structure, then apply the formula.
Can formal charge be positive or negative?
Yes. It can be positive, negative, or zero depending on electron distribution.
Why is formal charge important?
It helps determine the most stable molecular structure.
How do double bonds affect formal charge?
They increase bonding electrons, which are divided by 2 in the formula.
For More Visit Accurate Unit Convert












Leave a Reply